Drug metabolism
ROXY Reactors in Drug Metabolism
- Mimicking and predicting drug metabolism
- Complementary to in vivo and in vitro methods
- Fast: minutes vs. weeks or months of work
- Clean: no interferences with biological matrices
- Generating intermediates and reactive metabolites
In drug and xenobiotic metabolism the metabolites are formed instantaneously in the electrochemical cell mimicking the enzymatic biotransformation of the Cytochrome P450 reactions that usually take place in the liver (Phase I reaction). Hereby Electrochemistry becomes a truly biomimetic tool for enzymatic REDOX reactions.
Significant time and cost saving are possible compared to days or weeks using the traditional in vitro (e.g., microsomes) and/or in vivo methods (e.g., rodents, human, etc.). No cumbersome isolation form biological matrices (e.g., urine, plasma) is required and there is no longer risk of adsorption/binding to cells or other biological constituents. By adding glutathione after the electrochemical cell, adduct formation (phase II reaction) can be simulated and detected on-line by MS. Further advantages are: generation of intermediates and short-lived, unstable metabolites, no test animals (rodents), no biohazard risk (human liver microsomes).
For more details: see our technology page.
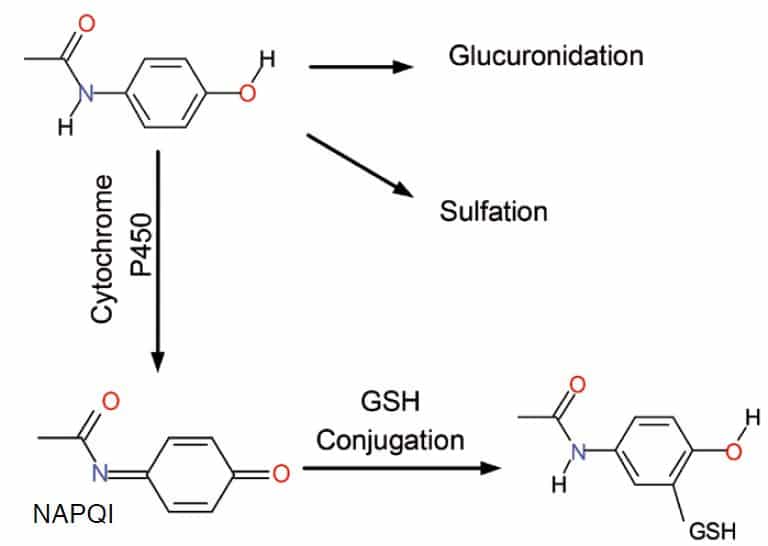
Metabolic pathways of acetaminophen. One of the metabolites conjugates with glutathione.
Electrochemical Reactors for MS and Synthesis
Environmental Degradation
Lipidomics, Lipid-Oxidation
Proteomics
Downloads
Posters